Sera Prognostics Reports Fourth Quarter 2021 Financial Results
Salt Lake City – March 29, 2022 – Sera Prognostics Inc., The Pregnancy Company® (Nasdaq: SERA), focused on improving maternal and neonatal health by providing innovative pregnancy biomarker information to doctors and patients, today announced financial results for the fourth quarter and full year 2021 ended December 31, 2021.
Recent Highlights:
- Our sales team is focusing geographically in high population density areas, where Anthem and other payers are concentrated.
- We continue discussions with large national, regional, self-insured employers and physician/healthcare networks who are motivated to make a positive clinical and economic difference in pregnancy. The recent announcement of the MultiPlan contract is an example of this and we anticipate additional future announcements as we successfully contract with multiple types of payers.
- Expanded the expertise of our Board of Directors through the appointment of highly respected technology, healthcare and financial leader, Sandra A. J. Lawrence, to our Board, bringing broad and profound leadership experience to Sera from having served on multiple public and private company boards.
- Announced that the Centers for Medicare & Medicaid Services (CMS) has set a Medicare payment rate of $750 for the existing unique Proprietary Laboratory Analysis (PLA) code, 0247U, for the PreTRM® Test, the company’s proprietary proteomic blood test for measuring a woman’s risk of spontaneous preterm birth to allow more timely intervention. The assigned rate further validates the commercial value of the PreTRM® Test.
- Announced publication of data confirming the performance of the IBP4/SHBG biomarker pair in diverse non-U.S. populations in a study funded by the Bill & Melinda Gates Foundation and in collaboration with AMANHI (Alliance for Maternal and Newborn Health Improvement), a multinational effort to improve health for pregnant and postpartum women and their babies in sub-Saharan Africa and South Asia. The study published in the peer-reviewed journal, Journal of Maternal-Fetal & Neonatal Medicine, demonstrated that our biomarkers can be used to predict spontaneous preterm birth (sPTB) in patient populations with baseline demographics and health care access that are distinct from those of U.S. populations.
- We recently presented data on Sera’s progress in improving preeclampsia prediction at the late-breaking poster session of the Society for Reproductive Investigation, demonstrating progress and the power of Sera’s proteomics and bioinformatics platform.
- Announced that the Company had been added to the NASDAQ Biotechnology Index® (Nasdaq: NBI) effective Monday, December 20, 2021.
“Fiscal year 2021 was a year full of notable developments and progress for Sera Prognostics as we commercially scale our business with the support of our shareholders, payors and employers who see the critical importance of our PreTRM® Test to improve lives and reduce ever expanding healthcare costs,” stated Gregory C. Critchfield, MD, MS, Chairman and CEO of Sera Prognostics. “While we have seen a slow initial rate of adoption, we expect to see growth during 2022 and beyond as COVID headwinds continue to wind down, as our sales staff make market inroads, and we accelerate our commercial activities. We believe that our commercial efforts will be benefited by new published data that further reinforce the significant value that our PreTRM® Test provides, as has been evidenced in a number of previously published study reports.”
Fourth Quarter 2021 Financial Results
Fourth quarter 2021 revenue of $26,000 compared to $6,000 for the same period of 2020.
Total operating expenses were $12.6 million, up from $4.8 million for the fourth quarter of 2020.
Research and development expenses for the fourth quarter of 2021 were $3.1 million compared to $2.1 million for the prior-year period due primarily to increased laboratory operations and clinical study costs.
Selling, general and administrative expenses for the fourth quarter of 2021 were $9.5 million, up from $2.7 million due primarily to increased headcount as the company has scaled its commercial operations and general corporate infrastructure, as well as increased costs related to operating as a public company following the Company’s initial public offering in July 2021.
Net loss for the fourth quarter of 2021 was $12.5 million compared to $5.4 million for the same quarter a year ago.
Full Year 2021 Financial Results
Total full year 2021 revenue of $82,000 compared to $25,000 for full year 2020.
Total operating expenses were $35.5 million, up from $18.0 million for 2020.
Research and development expenses for 2021 were $11.0 million compared to $7.8 million for the prior year due to increased laboratory operations and clinical study costs.
Selling, general and administrative expenses for 2021 were $24.4 million, up from $10.2 million for the prior year due primarily to the aforementioned increased headcount and corporate infrastructure expenditures and costs related to operating as a public company.
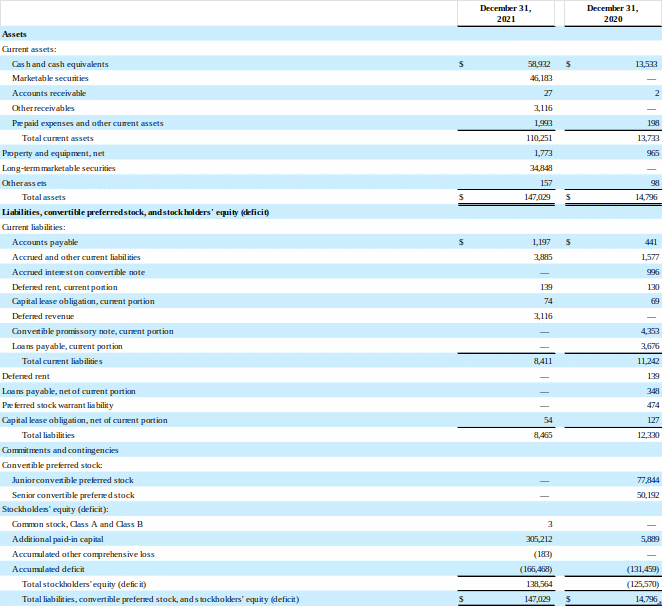
As of December 31, 2021, the Company had cash and cash equivalents of approximately $58.9 million and $81.0 million of available-for-sale securities.
Conference Call Information
Sera Prognostics will host a corresponding conference call and live webcast today to discuss fourth quarter 2021 operational highlights, financial results and key topics at 5:00 p.m. Eastern Time. Individuals interested in listening to the conference call may do so by dialing the following:
US domestic callers: (877) 870-4263
International callers: (412) 317-0790
Webcast Registration Link: https://www.webcaster4.com/Webcast/Page/2809/44889
Live audio of the webcast will be available online from the Investors page of the Company’s website at www.seraprognostics.com. The webcast will be archived on the Investors page and will be available for one year.
About Sera Prognostics, Inc.
Sera Prognostics is a leading health diagnostics company dedicated to improving the lives of women and babies through precision pregnancy care. Sera’s mission is to deliver early, pivotal information in pregnancy to physicians, enabling them to improve the health of their patients, resulting in reductions in the costs of healthcare delivery. Sera has a robust pipeline of innovative diagnostic tests focused on the early prediction of preterm birth risk and other complications of pregnancy. Sera’s precision medicine PreTRM® Test reports to a physician the individualized risk of spontaneous premature delivery in a pregnancy, enabling earlier proactive interventions in women with higher risk. Sera Prognostics is located in Salt Lake City, Utah.
About Preterm Birth
Preterm birth is defined as any birth before 37 weeks’ gestation and is the leading cause of illness and death in newborns. The 2021 March of Dimes Report Card shows that more than one in ten infants is born prematurely. Prematurity is associated with a significantly increased risk of major long-term medical complications, including learning disabilities, cerebral palsy, chronic respiratory illness, intellectual disability, seizures, and vision and hearing loss, and can generate significant costs throughout the lives of affected children. The annual health care costs to manage short- and long-term complications of prematurity in the United States were estimated to be approximately $25 billion for 2016.
About the PreTRM® Test
The PreTRM® test is the only broadly validated, commercially available blood-based biomarker test that provides an early, accurate and individualized risk prediction for spontaneous preterm birth in asymptomatic singleton pregnancies. The PreTRM® test measures and analyzes proteins in the blood that are highly predictive of preterm birth. The PreTRM® test permits physicians to identify, during the 19th or 20th week of pregnancy, which women are at increased risk for preterm birth, enabling more informed, personalized clinical decisions based on each woman’s individual risk. The PreTRM® test is ordered by a medical professional.
Sera Prognostics, the Sera Prognostics logo, The Pregnancy Company, and PreTRM are trademarks or registered trademarks of Sera Prognostics, Inc. in the United States and/or other countries.
Safe Harbor Statement
This press release contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995, including statements relating to anticipated additional future announcements of successful contracts with multiple types of payers; expected growth during 2022; sales staff making market inroads; accelerating commercial activities; the Company’s commercial efforts benefitting from new published data to further reinforce the significant value that the PreTRM® Test provides; the Company’s strategy, future operations, prospects, plans, objectives of management; the Company’s expectations regarding the commercialization of the PreTRM® Test; the Company’s planned conference call and live webcast to discuss the quarter’s operational highlights, financial results and key topics; availability of audio of the webcast online from the Investors page of the Company’s website; and the company’s strategic directives under the caption “About Sera Prognostics, Inc.” These “forward-looking statements” are based on management’s current expectations of future events and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by forward-looking statements. These risks and uncertainties include, but are not limited to: net losses, cash generation, and the potential need to raise more capital; revenues from the PreTRM test representing substantially all Company revenues to date; the need for broad scientific and market acceptance of the PreTRM test; a concentrated number of material customers; our ability to introduce new products; potential competition; our proprietary biobank; critical suppliers; the ongoing COVID-19 pandemic and its impact on our operations, as well as the business or operations of third parties with whom we conduct business; estimates of total addressable market opportunity and forecasts of market growth; potential third-party payer coverage and reimbursement; new reimbursement methodologies applicable to the PreTRM test, including new CPT codes and payment rates for those codes; changes in FDA regulation of laboratory-developed tests; the intellectual property rights protecting our tests and market position; and other factors discussed under the heading “Risk Factors” contained in our Quarterly Reports on Form 10-Q, Annual Reports on Form 10-K, or Current Reports on Form 8-K filed with the SEC from time to time. All information in this press release is as of the date of the release, and the Company undertakes no duty to update this information unless required by law.
Investor Contact
Peter DeNardo, CapComm Partners
peter@capcommpartners.com
+1 (415) 389-6400
Media Contact
Erich Sandoval, Lazar FINN
Erich.sandoval@finnpartners.com
+1 (917) 497-2867
SERA PROGNOSTICS, INC.
Statements of Operations
(unaudited)
(in thousands, except share and per share data)
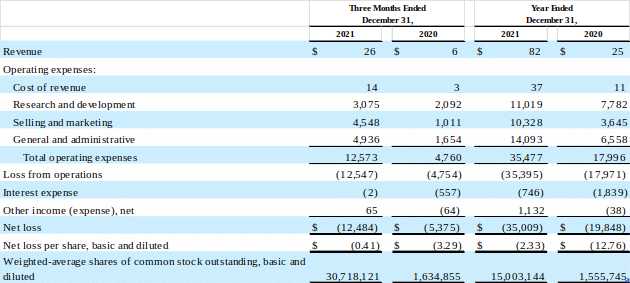
SERA PROGNOSTICS, INC.
Balance Sheets
(unaudited)
(in thousands)